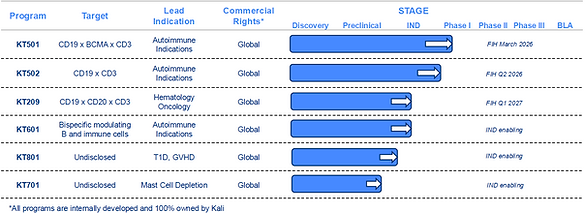
KT501 - CD19xBCMAxCD3
KT501 is a novel IgG-like tri-specific T cell engager, designed to bind CD3, CD19, BCMA with high affinity, thereby targeting a broad range of B cell populations. Utilizing an innovative molecular design aimed at maximizing potency while minimizing cytokine release by a unique CD3 masking platform. KT501 has demonstrated robust B cell cytotoxicity and reduced cytokine production in vitro and in non-human primate (NHP) due to its cross activity in NHP.
Kali Therapeutics is initiating a first-in-human study for KT501 in Rheumatoid arthritis (RA) in December 2025 and is expanding KT501 into various autoimmune indications.

The antibody format in the image is for illustration purposes only. KT501 is of a different format.
KT502 - CD19xCD3
KT502 was selected on an innovative molecular design that maximizes B cell cytotoxicity while minimizes cytokine release through a unique CD3 masking technology. KT502 has demonstrated robust B cell cytotoxicity and reduced cytokine production in vitro and in non-human primate (NHP) due to its cross activity in NHP.
IND-enabling activities are currently underway for KT502, with a first-in-human study to be initiated in 1H 2026.
KT209 – CD19xCD20xCD3
KT209 is a next-generation tri-specific T cell engager designed to target both CD19 and CD20, providing comprehensive dual-antigen coverage across a broad range of B-cell malignancies. Built on the same innovative CD3 masking platform as KT501 and KT502, KT209 is engineered to achieve a superior therapeutic index by decoupling potent B-cell cytotoxicity from systemic cytokine release.
In preclinical evaluations, KT209 has demonstrated exceptional anti-tumor activity, exhibiting 100- to 1,000-fold greater potency in killing multiple different B-cell lymphoma tumor cell lines compared to currently approved CD20xCD3 or CD19xCD3 therapies. The program has been proven safe in non-human primate (NHP) studies, demonstrating favorable transit and minimal cytokine release, confirming the platform’s ability to maintain a robust safety profile even at extreme levels of potency.
IND-enabling activities are currently underway for KT209, with a first-in-human study to be initiated in 2H 2026.

Ramírez-Valle, F., Maranville, J.C., Roy, S. et al. Sequential immunotherapy: towards cures for autoimmunity. Nat Rev Drug Discov (2024). https://doi.org/10.1038/s41573-024-00959-8
.png)
